
Therapeutic strategies for DMD and age-associated neuromuscular disorders
Keywords: Muscle metabolism, muscular dystrophy, aging, therapeutic strategies
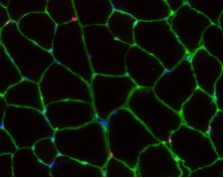
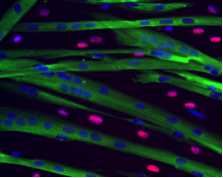
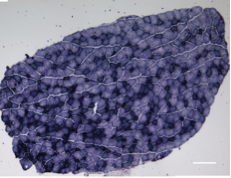
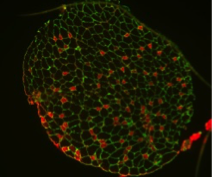
A long-standing research interest in our lab has been focused on the crosstalk between cell cycle regulation and the differentiation program of skeletal muscle stem cells, with specific attention to the role of D-type cyclins in these processes. In past years our studies have revealed a unique function played by cyclin D3 in the control of the developmental program of skeletal muscle precursor cells both in vitro and in vivo. Recently, we have uncovered a novel cell cycle-independent role for cyclin D3 in regulating the contractile and metabolic properties of skeletal muscle fibers and whole-body energy metabolism. Furthermore, we have demonstrated that genetic inactivation of cyclin D3 in the mdx mouse model of Duchenne Muscular Dystrophy (DMD) ameliorates the disease phenotype, by promoting a slower, more oxidative profile of muscle fibers that is more resistant to dystrophic pathology.
- In view of a possible translational approach, we are now evaluating the therapeutic efficacy of targeting cyclin D3 activity in adult mdx mice when the signs of disease are already evident.
A related research interest is centered on the regulatory crosstalk between skeletal muscle and brain. Skeletal muscle is the most abundant tissue of the body and a major metabolic organ. Furthermore, this tissue produces and secretes growth factors and cytokines (commonly termed myokines) that modulate systemic physiology. Skeletal muscle may crosstalk with brain via direct muscle-nerve interaction and release of metabolites and myokines in response to muscle contraction, diet-derived nutrients, and stress. There is evidence that myokines and other factors secreted by skeletal muscle contribute to the regulation of neurogenesis, memory and learning and that chronic muscle disorders can compromise hippocampal function.
- Our ongoing studies aim to evaluate the potential positive impact of external stimuli that influence pathways involved in energy metabolism (natural bioactive dietary compounds, exercise etc) on muscle dysfunctions and hippocampal neurogenesis defects in mouse models of muscular dystrophy and aging.
IBBC Collaborators: Laura Micheli, Felice Tirone, Siro Luvisetto
Education:
- 1973 Graduated from High School
- 1979 Degree in Biological Sciences (cum laude), University of Naples “Federico II°”, Italy.
Positions:
- 1980 Post-doctoral fellow, Unitè de Genie Genetique, Institut Pasteur, Paris (France)
- 1981-1982 Post-doctoral fellow, Department of Microbiology, University of Chicago, Chicago (USA)
- 1982-1984 Post-doctoral fellow, Department of Pathology, New York University School of Medicine, New York (USA)
- 1984-1985 Research fellow, National Research Council, Institute of Genetics and Biophysics, Napoli (Italy)
- 1985-1990 Research fellow, University “La Sapienza”, Department of Human Biopathology, Roma (Italy)
- 1990-2001 Research Scientist – National Research Council, Institute of Cell Biology, Roma, Italy
- 2001-present Senior Research Scientist – National Research Council, Institute of Neurobiology and Moleculare Medicine, Institute of Cell Biology and Neurobiology and Institute of Biochemistry and Cell Biology Roma, Italy
Awards:
1979: “Ernesto Scoffone and Terenzio Cremona” Thesis Award, sponsored by the Italian Society of Biophysics and Molecular Biology
1980: Research Fellowship Award from Fondazione “Istituto Pasteur-Fondazione Cenci- Bolognetti”
Teaching and Professional activities:
2005-2013 Teaching Board Member, PhD School in “Genetics and Cellular Biology”, Tuscia University, Viterbo, Italy
2013 Member of Grant Review Committee “Future in Research 2013”. Ministry of University and Research (MIUR)
2014 Member of Grant Review Committee “SIR” (Scientific Independence of young Researchers). Ministry of University and Research (MIUR)
Ad hoc peer reviewer for several scientific journals, including Stem Cells, Cell Death Differentiation, Experimental Cell Research, Journal of Cellular Biochemistry, Biochemistry and Cell Biology
Micheli L, Bertini L, Bonato A, Villanova N, Caruso C, Caruso M, Bernini R, Tirone F. (2023) Role of Hydroxytyrosol and Oleuropein in the Prevention of Aging and Related Disorders: Focus on Neurodegeneration, Skeletal Muscle Dysfunction and Gut Microbiota. Nutrients. 15:1767. doi: 10.3390/nu15071767.
Bonato A, Raparelli G, Luvisetto S, Forconi F, Cosentino M, Tirone F, Rizzuto E, Caruso M. (2023) Cyclin D3 deficiency promotes a slower, more oxidative skeletal muscle phenotype and ameliorates pathophysiology in the mdx mouse model of Duchenne muscular dystrophy. FASEB J. 37:e23025. doi: 10.1096/fj.202201769R.
Belli R, Bonato A, De Angelis L, Mirabilii S, Ricciardi MR, Tafuri A, Molfino A, Leigheb M, Costelli P, Caruso M, Muscaritoli M and Ferraro E. (2019) Metabolic Reprogramming Promotes Myogenesis During Aging. Frontiers in Physiology. 10:897. doi: 10.3389/fphys.2019.00897.
Giannattasio S, Giacovazzo G, Bonato A, Caruso C, Luvisetto S, Coccurello R and Caruso M. (2018) Lack of cyclin D3 induces skeletal muscle fiber-type shifting, increased endurance performance and hypermetabolism. Scientific Reports. 8:12792.doi: 10.1038/s41598-018-31090-5
Albini, S., Coutinho Toto, P., Dall’Agnese, A., Malecova, B., Cenciarelli, C., Felsani, A., Caruso, M., Bultman, S.J., Puri, PL. (2015) Brahma is required for cell cycle arrest and late muscle gene expression during skeletal myogenesis. EMBO Rep., 16:1037-50. doi: 10.15252/embr.201540159).
De Luca G, Ferretti R, Bruschi M, Mezzaroma E and Caruso M. (2013) Cyclin D3 critically regulates the balance between self-renewal and differentiation in skeletal muscle stem cells. Stem Cells 31: 2478-2491.
Orcid: http://orcid.org/0000-0001-5445-6343
Post-doc:
Agnese Bonato
bonato.agnese@gmail.com
CNR Research fellow
Giada Raparelli
giadaraparelli@icloud.com
2024-2028: Associazione per le Neuroscienze Giuseppe Moruzzi. “Attivazione mediante stimoli neurogenici delle cellule staminali neurali nelle nicchie neurogeniche adulte e nel cervelletto: studio della fisiopatologia correlata e del ruolo dell’asse cervello-muscolo”, Role: Partner
2023-2025: (CNR-FOE 2021)/ Progetto Nutrage Attività IBBC-5 “Effetto dei polifenoli dell’olio di oliva sulle cellule staminali delle nicchie neurogeniche e muscolari e sul microbiota intestinale in modelli murini di invecchiamento”, Role: Partner
2020-2023: Duchenne Parent Project, Netherlands project N. 19018 “Evaluation of cyclin D3 as a potential target to remodel dystrophic muscle toward the slow, oxidative phenotype”, Role: Principal investigator
2020-2024: Lazio Innova POR FESR LAZIO Programmazione 2014 – 2020, Project N. A0375-2020-36407 coordinated project “Declino cognitivo, perdita di funzionalità muscolare e alterazioni del microbiota intestinale associati all¹invecchiamento: effetti protettivi dell¹idrossitirosolo, componente dell¹olio di oliva e di estratti da sottoprodotti oleari”, Role: Partner
18/10/2018. Ciclina D3: la sua assenza rende resistenti alla fatica muscolare.
La mancanza di questa proteina fa prevalere le fibre muscolari che si contraggono lentamente su quelle a contrazione più rapida. Gli animali che ne sono sprovvisti hanno maggior resistenza all’affaticamento ed un maggiore dispendio energetico, con conseguenze sul metabolismo. Ad indicarlo uno studio dell’Istituto di biologia cellulare e neurobiologia del Cnr pubblicato su Scientific Reports
