Group Leader: Francesca Ruberti
Topic : Neurobiology
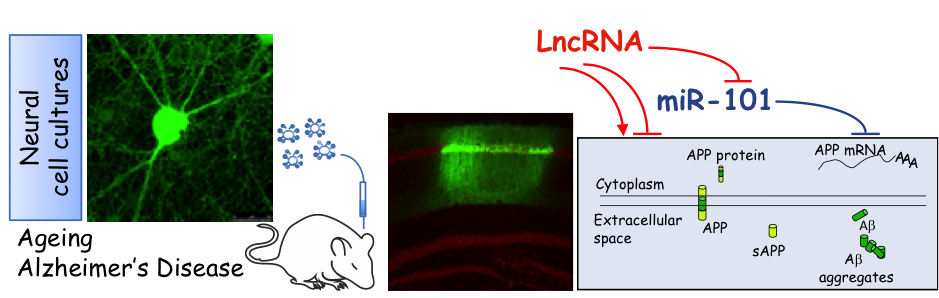
Non-coding RNAs and in particular microRNAs tunes brain cell differentiation and synaptic development and modulates brain functions such as synaptic plasticity, memory formation and behavioral performance.
Our research aims to identify long noncoding RNAs (lncRNAs) and/or microRNAs as well as their interactions, and to investigate their roles in neuronal and glia cells during physiological and pathological conditions. Main focus is the study of these molecules in Ageing and Alzheimer’s Disease (AD).
Primary rodent neural cell cultures, cell lines, human neuronal and glia cells differentiated from iPSC (induced pluripotent stem cells)-derived NSC (neural stem cells) as well as mouse models are instrumental to our studies. Previous findings in our laboratory have shown that miR-101 regulates directly AD-related genes, Amyloid precursor protein (APP) and Ranbp9, in rodent hippocampal neurons in vitro and in vivo (Vilardo et al., 2010; Barbato et al., 2014). More recently we found that inhibition of miR-101post-transcriptional regulation in CA1 hippocampal neurons of adult C57BL/SJ mice, by stereotaxic injection of a lentiviral miRNA sponge, leads to cognitive decline (Barbato et al., submitted). We are now characterizing the molecular pathways involved in hippocampal dependent cognitive decline after miR-101downregulation that may occur during aging and AD. Furthermore, we are searching for lncRNAs that might regulate miR-101 and/or other microRNAs and pathways associated to Ageing/AD
Key Pubblications
Cognitive decline and modulation of Alzheimer’s disease-related genes after inhibition of microRNA-101 in mouse hippocampal neurons Barbato C, Giacovazzo G, Albiero F, Scardigli R, Scopa C, Ciotti MT, Strimpakos G, Coccurello R, Ruberti F (submitted)
MicroRNA Biology and Function in the Nervous System Barbato C and Ruberti F (2016) in “Mapping Nervous System Diseases via MicroRNAs” (2016) (eds. Barbato C and Ruberti F) by CRC Press, Pages 3-18, ISBN 9781482263527- cat K24373. Series: Frontiers in Neurotherapeutics.
MicroRNA Landscape in Alzheimer’s Disease. Cogoni C, Ruberti F. and Barbato C (2015) CNS Neurol Disord Drug Targets 14(2):168-75.
Selective inhibition of miR-92 in hippocampal neurons alters contextual fear memory. Vetere G, Barbato C, Pezzola S, Frisone P, Aceti M, Ciotti M, Cogoni C, Ammassari-Teule M, Ruberti F. (2014) Hippocampus. 24:1458-65. doi: 10.1002/hipo.22326.
A lentiviral sponge for miR-101 regulates RanBP9 expression and amyloid precursor protein metabolism in hippocampal neurons. Barbato C, Pezzola S, Caggiano C, Antonelli M, Frisone P, Ciotti MT and Ruberti F (2014) Frontiers in Cellular Neuroscience 8:37. doi: 10.3389/fncel.2014.00037
MicroRNA-101 regulates amyloid precursor protein expression in hippocampal neurons. Vilardo E, Barbato C, Ciotti MT, Cogoni C and Ruberti F (2010) J. Biol Chem 285:18344-18351.
Research group
Researchers:
Dr. Christian Barbato
Dr. Roberto Coccurello roberto.coccurello@cnr.it
Associated Scientists:
Prof. Nadia Canu nadia.canu@uniroma2.it
Program and resources
PNR-CNR Aging Program 2012-2016
“Study of microRNAs in Alzheimer’s disease pathogenesis and progression”.
Role: Principal Investigator
Progetto Bandiera Nanomax 2012-2014
Nadine, Workpackage 8 “Serum miRNAs in Alzheimer’s Disease”
Role: Coinvestigator
3886 SD/sd 2008.2404 Programma Neuroscienze Barbato (PI) 9/01/09-9/01/11
‘MIND: MicroRNA In Neurodegenerative Diseases’
This study is dedicated to analyze the expression of microRNAs in human Alzheimer’s disease brains.
Role: Coinvestigator
RSTL.059.012 –CNR grant- Study of APP regulation by microRNAs and implication in Alzheimer’s Disease 01/2008-12/2009.
Role: Principal Investigator
“Fondo speciale per le Neuroscienze”-Regione Lazio-FILAS SpA 2005-2007
Role: Co-Investigator
